Sydney, Australia 20 June 2023
Clarity Pharmaceuticals (ASX: CU6) (“Clarity”), a clinical stage radiopharmaceutical company with a mission to develop next-generation products that improve treatment outcomes for children and adults with cancer, is pleased to announce it has commenced its 64Cu/67Cu SAR-Bombesin Phase I/II trial in metastatic castrate resistant prostate cancer (mCRPC) with the opening of the first site at BAMF Health, Inc in Michigan.
COMBAT (Copper-67 SAR Bombesin in metastatic castrate resistant prostate cancer, NCT05633160)1 is a dose escalation and cohort expansion trial for up to 38 participants. The aim for the trial is to determine the safety and efficacy of 67Cu-SAR-Bombesin in participants with gastrin-releasing peptide receptor (GRPr) expressing mCRPC in patients who are ineligible for therapy with 177Lu PSMA-617.
Clarity’s Executive Chairman, Dr Alan Taylor, commented, “We are excited to initiate our first theranostic clinical trial of the SAR-Bombesin product. COMBAT is our third theranostic trial in the US and a second theranostic trial in prostate cancer, an indication that continues to have a high unmet need. In the VISION trial for the recently approved 177Lu PSMA-617, median overall survival increased from approximately 11 months on standard of care therapy to approximately 15 months with 177Lu PSMA-617 plus standard of care. Despite this, around a quarter of men with mCRPC do not have PSMA-expressing lesions, making it impossible to offer PSMA-targeted products as therapy for this group of patients. As a very large proportion of prostate cancers express GRPr, SAR-Bombesin is an exciting new prospect for better treating these patients that have few therapeutic options at present in the face of a devastating diagnosis.
“We look forward to progressing the COMBAT trial and building on the compelling data from our preclinical studies as well as from three diagnostic trials with this product. SAR-Bombesin has already resulted in improvements to the management of prostate cancer for patients with PSMA-negative or low PSMA expressing tumours and we hope to confirm its safety and efficacy in the theranostic trial.”
Overview of Clarity’s Prostate Cancer Clinical Trial Program
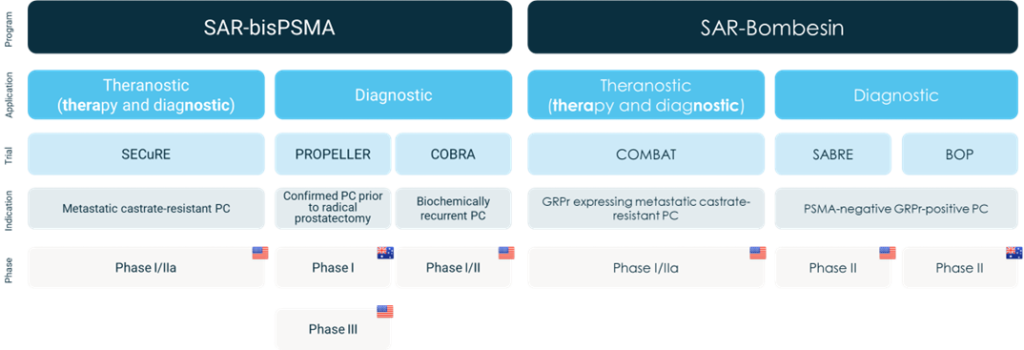
About SAR-Bombesin
SAR-Bombesin is a highly targeted pan-cancer radiopharmaceutical with broad cancer application. It targets the gastrin-releasing peptide receptor (GRPr) present on cells of a range of cancers, including but not limited to prostate, breast and ovarian cancers. GRPr is found in up to 100% of prostate cancers, including prostate cancers that don’t express PSMA (PSMA-negative)2-6. The product utilises Clarity’s proprietary sarcophagine (SAR) technology that securely holds copper isotopes inside a cage-like structure, called a chelator. Unlike other commercially available chelators, the SAR technology prevents copper leakage into the body. SAR-Bombesin is a Targeted Copper Theranostic (TCT) that can be used with isotopes of copper-64 (Cu-64 or 64Cu) for imaging and copper-67 (Cu-67 or 67Cu) for therapy.
About Prostate Cancer
Prostate cancer is the second most common cancer diagnosed in men globally and the fifth leading cause of cancer death worldwide7. The American Cancer Institute estimates in 2023 there will be 288,300 new cases of prostate cancer in the US and around 34,700 deaths from the disease8.
Approximately 25% of mCRPC patients have low or no uptake of a PSMA-targeting tracer.9 These patients are therefore unlikely to respond to therapeutic PSMA-targeted products and currently have few treatment options available to them. Given the prostate cancer indication is one of the largest in oncology, there is a significant unmet medical need in this segment. The SAR-Bombesin product could offer valuable imaging and therapeutic options for not only PSMA-negative patients, but also the large number of patients that have the target receptor on their cancers.
About Clarity
Clarity is a clinical stage radiopharmaceutical company focused on the treatment of serious disease. The Company is a leader in innovative radiopharmaceuticals, developing targeted copper theranostics based on its SAR Technology Platform for the treatment of cancer in children and adults.
www.claritypharmaceuticals.com
References
- Clinicaltrials.gov NCT05633160, https://www.clinicaltrials.gov/ct2/show/NCT05633160
- Markwalder R, Reubi JC. Gastrin-releasing peptide receptors in the human prostate: relation to neoplastic transformation. Cancer research. 1999;59(5):1152-1159.
- Fleischmann A, Waser B, Reubi JC. High expression of gastrin-releasing peptide receptors in the vascular bed of urinary tract cancers: promising candidates for vascular targeting applications. Endocrine-related cancer. 2009;16(2):623-633.
- Ananias HJ, van den Heuvel MC, Helfrich W, de Jong IJ. Expression of the gastrin-releasing peptide receptor, the prostate stem cell antigen and the prostate-specific membrane antigen in lymph node and bone metastases of prostate cancer. The Prostate. 2009;69(10):1101-1108.
- Reubi JC, Wenger S, Schmuckli-Maurer J, Schaer JC, Gugger M. Bombesin receptor subtypes in human cancers: detection with the universal radioligand (125)I-[D-TYR(6), beta-ALA(11), PHE(13), NLE(14)] bombesin(6-14). Clin Cancer Res. 2002;8(4):1139-1146.
- Sun B, Halmos G, Schally AV, Wang X, Martinez M. Presence of receptors for bombesin/gastrin-releasing peptide and mRNA for three receptor subtypes in human prostate cancers. The Prostate. 2000;42(4):295-303.
- Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries, https://acsjournals.onlinelibrary.wiley.com/doi/10.3322/caac.21660
- American Cancer Society: Cancer Statistics Center, https://cancerstatisticscenter.cancer.org/?_ga=2.79808020.284532473.1620009137-1916069442.1615761164#!/cancer-site/Prostate
- Vlachostergios PJ, Niaz MJ, Sun M, et al. Prostate-Specific Membrane Antigen Uptake and Survival in Metastatic Castration-Resistant Prostate Cancer. Frontiers in oncology. 2021;11.
Media Contact
Clarity Pharmaceuticals
Dr Alan Taylor
Executive Chairman
+61 (0)413 871 165
ataylor@claritypharm.com
Citadel-MAGNUS
Catherine Strong
Investor/Media Relations
+61 (0)406 759 268
cstrong@citadelmagnus.com
This announcement has been authorised for release by the Executive Chairman.
