Sydney, Australia 26 November 2024
Clarity Pharmaceuticals (ASX: CU6) (“Clarity”), a clinical-stage radiopharmaceutical company with a mission to develop next-generation products that improve treatment outcomes for children and adults with cancer, is pleased to announce that two participants have been dosed and imaged days after the commencement of the Co-PSMA Investigator-Initiated Trial (IIT). The study is evaluating the performance of Clarity’s diagnostic product, 64Cu-SAR-bisPSMA, in comparison to standard-of-care (SOC) 68Ga-PSMA-11 for the detection of prostate cancer recurrence. No safety issues were observed during the administration of 64Cu-SAR-bisPSMA.
Co-PSMA, derived from “Comparative performance of 64Copper [64Cu]-SAR-bisPSMA vs 68Ga-PSMA-11 PET CT for the detection of prostate cancer recurrence in the setting of biochemical failure following radical prostatectomy,” is a prospective, Phase II comparative imaging trial in 50 patients with biochemical recurrence (BCR) post-radical prostatectomy who are being considered for curative salvage radiotherapy, led by Prof Louise Emmett at St Vincent’s Hospital, Sydney. The primary objective of the study is to compare the detection rate of sites of prostate cancer recurrence, as determined by number of lesions per patient, between 64Cu-SAR-bisPSMA and 68Ga-PSMA-11 positron emission tomography (PET)/computed tomography (CT).
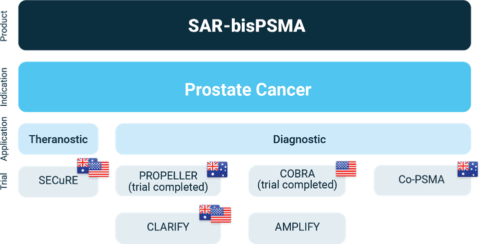
The diagnostic capabilities of 64Cu-SAR-bisPSMA compared to SOC diagnostic imaging have been demonstrated in two prospective clinical trials, PROPELLER and COBRA. The PROPELLER study, conducted in pre-radical prostatectomy patients, showed 2-3 times higher tumor uptake and contrast, and the detection of additional prostate cancer lesions with 64Cu-SAR-bisPSMA compared to 68Ga-PSMA-111 using same day imaging only (Figures 1 and 2).
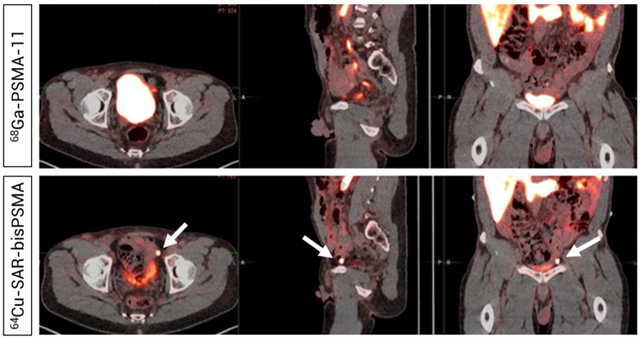
Figure 1: Detection of prostate cancer in pelvic lymph node by 64Cu-SAR-bisPSMA, but not by 68Ga-PSMA-11 PET/CT. Readers did not detect uptake in pelvic lymph nodes on the 68Ga-PSMA-11 PET/CT (Top). PET/CT demonstrated uptake of 64Cu-SAR-bisPSMA in a left pelvic lymph node according to both Readers and prostate cancer was confirmed via histopathology. Arrows highlight the node detected on 64Cu-SAR-bisPSMA PET/CT. Interval between serial imaging: 7 days.
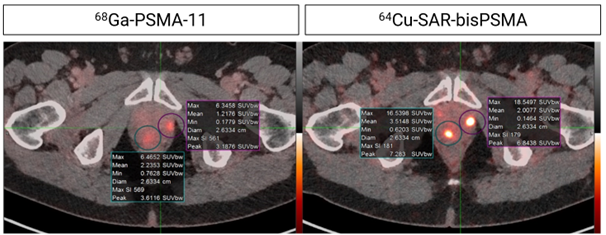
Figure 2: Higher uptake and contrast in lesions identified by 64Cu-SAR-bisPSMA compared to 68Ga-PSMA-11. Concordant lesions on 64Cu-SAR-bisPSMA and 68Ga-PSMA-11 PET/CT consistently showed 2-3 times higher maximum and mean standardised uptake value (SUVmax, SUVmean) and tumour-to-background ratio (TBR) with 64Cu-SAR-bisPSMA compared to 68Ga-PSMA-11 (statistically significant values for all parameters). Interval between scans: 8 days.
The COBRA study, conducted in patients with BCR of prostate cancer who had a negative or equivocal SOC scan, showed that 82% more lesions were identified by 64Cu-SAR-bisPSMA on next-day imaging (average across 3 readers) compared to same-day imaging2. Delayed imaging also showed that lesions detected on next-day imaging had a higher tracer uptake and contrast vs. same-day imaging, as well as allowing for the identification of lesions under 5 millimeters (mm) in size (Figure 3). In this study, 64Cu-SAR-bisPSMA identified lesions of <5 mm in size in 14% of participants.
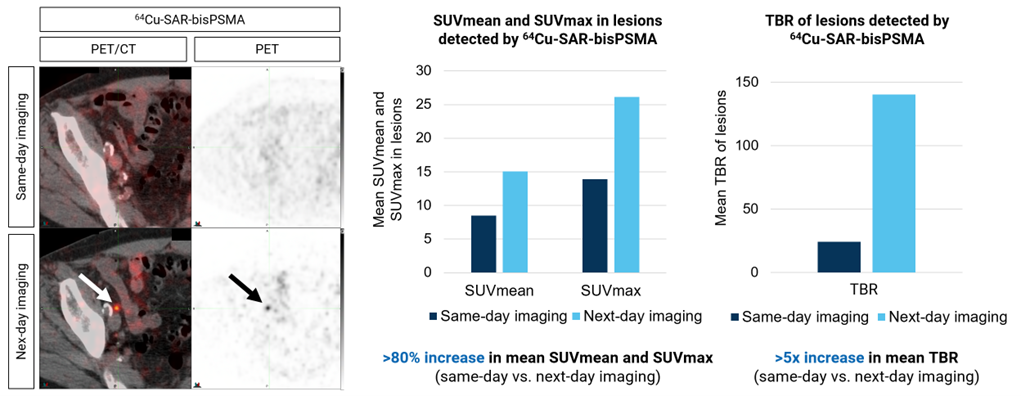
Figure 3: Detection of a lesion in the 2 mm range and higher uptake and contrast in lesions on next-day vs. same-day imaging using 64Cu-SAR-bisPSMA. Left (images): Right pelvic lymph node showing uptake of 64Cu-SAR-bisPSMA on next-day imaging (arrows, bottom images). Lesion size: 1.9 mm x 2.6 mm. SUVmean 8.0, SUVmax 8.2 and TBR 67.9. Right (graphs): SUVmean/max and TBR comparing same-day and next-day imaging. Average increase across 3 readers. The SUVmax, SUVmean and TBR were assessed in up to 25 lesions per patient on each 64Cu-SAR-bisPSMA scan.
Dr Alan Taylor, Executive Chairperson of Clarity Pharmaceuticals, commented: “We are very excited to help some men who have recurrent disease based on rising prostate specific antigen (PSA) levels following radical prostatectomy in our own city of Sydney through supporting this Co-PSMA trial. We are pleased to see the commencement of this Co-PSMA trial with a world-renowned thought leader in the theranostics space, Prof Louise Emmett, at one of the most prominent hospitals in the country, St Vincent’s Hospital Sydney. Patients have already been dosed and had same-day and next-day imaging within a few days after Co-PSMA initiation. This indicates that there is a high unmet need for improved prostate cancer diagnostics in this patient population, which is the largest population for PSMA imaging globally. The diagnostic capabilities of 64Cu-SAR-bisPSMA compared to SOC diagnostic imaging have been demonstrated in two prospective clinical trials, PROPELLER and COBRA. Furthermore, the differences between 64Cu-SAR-bisPSMA and SOC PSMA PET agents were observed even when state-of-the-art whole body PET cameras were used.
“Further to this trial, Clarity continues to progress our 2 registrational Phase III trials, CLARIFY and AMPLIFY, in the U.S. and Australia. Due to the high-volume centralised manufacture of product with no reliance on short half-life isotopes, we do not anticipate any issues of recruitment with any of the ongoing and planned trials with 64Cu-SAR-bisPSMA.
“Knowing where the cancer is located is essential for clinicians to decide what the best treatment is for each patient. As the diagnostic performance of 64Cu-SAR-bisPSMA has been demonstrated through previous clinical trials, such as COBRA and PROPELLER, we eagerly await the results of this head-to-head comparison between 64Cu-SAR-bisPSMA and 68Ga-PSMA-11 PET in the hope of opening the opportunity for earlier detection of disease as we progress towards our ultimate goal of better treating people with cancer.”
About SAR-bisPSMA
SAR-bisPSMA derives its name from the word “bis”, which reflects a novel approach of connecting two PSMA-targeting agents to Clarity’s proprietary sarcophagine (SAR) technology that securely holds copper isotopes inside a cage-like structure, called a chelator. Unlike other commercially available chelators, the SAR technology prevents copper leakage into the body. SAR-bisPSMA is a Targeted Copper Theranostic (TCT) that can be used with isotopes of copper-64 (Cu-64 or 64Cu) for imaging and copper-67 (Cu-67 or 67Cu) for therapy.
64Cu-SAR-bisPSMA is an unregistered product. The safety and efficacy of 64Cu-SAR-bisPSMA has not been assessed by health authorities such as the US Food and Drug Administration (FDA) or the Therapeutic Goods Administration (TGA). There is no guarantee that this product will become commercially available. Among 82 patients who received 64Cu-SAR-bisPSMA in PROPELLER and COBRA, 2 adverse reactions were reported in 2 participants (mild occasional metallic taste and moderate worsening of type II diabetes, both resolved)1,2.
Overview of Clarity’s SAR-bisPSMA clinical program
About Prostate Cancer
Prostate cancer is the second most common cancer diagnosed in men globally and the fifth leading cause of cancer death in men worldwide3. Prostate cancer is the second-leading causes of cancer death in American men. The American Cancer Institute estimates in 2024 there will be 299,310 new cases of prostate cancer in the US and around 35,250 deaths from the disease4.
About Clarity Pharmaceuticals
Clarity is a clinical stage radiopharmaceutical company focused on the treatment of serious disease. The Company is a leader in innovative radiopharmaceuticals, developing targeted copper theranostics based on its SAR Technology Platform for the treatment of cancer in children and adults.
References
- Lengyelova et al. 64Cu-SAR-bisPSMA (PROPELLER) positron emission tomography (PET) imaging in patients with confirmed prostate cancer. ASCO, 2023.
- Nordquist et al. COBRA: Assessment of safety and efficacy of 64Cu-SAR-bisPSMA in patients with biochemical recurrence of prostate cancer following definitive therapy. EANM, 2024.
- Global Cancer Statistics 2022: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries, https://acsjournals.onlinelibrary.wiley.com/doi/10.3322/caac.21834
- American Cancer Society: Key Statistics for Prostate Cancer, https://www.cancer.org/cancer/prostate-cancer/about/key-statistics.html
- Data on file.
- Shore et al. Biochemical recurrence in patients with prostate cancer after primary definitive therapy: treatment based on risk stratification. Prostate Cancer Prostatic Dis. 2024 Jun;27(2):192-201. doi: 10.1038/s41391-023-00712-z.
- Falkenbach et al. Size of lymph-node metastases in prostate cancer patients undergoing radical prostatectomy: implication for imaging and oncologic follow-up of 2705 lymph-node positive patients. World J Urol. 2024 Jan 20;42(1):38. doi: 10.1007/s00345-023-04724-1.
- Krasnovskaya et al. Recent Advances in 64Cu/67Cu-Based Radiopharmaceuticals. Int J Mol Sci. 2023 May 23;24(11):9154. doi: 10.3390/ijms24119154.
Media Contacts
Clarity Pharmaceuticals
Dr Alan Taylor
Executive Chairperson
ataylor@claritypharm.com
Sodali
Catherine Strong
Investor/Media Relations
+61 (0)406 759 268
catherine.strong@sodali.com
This announcement has been authorised for release by the Executive Chairperson.
